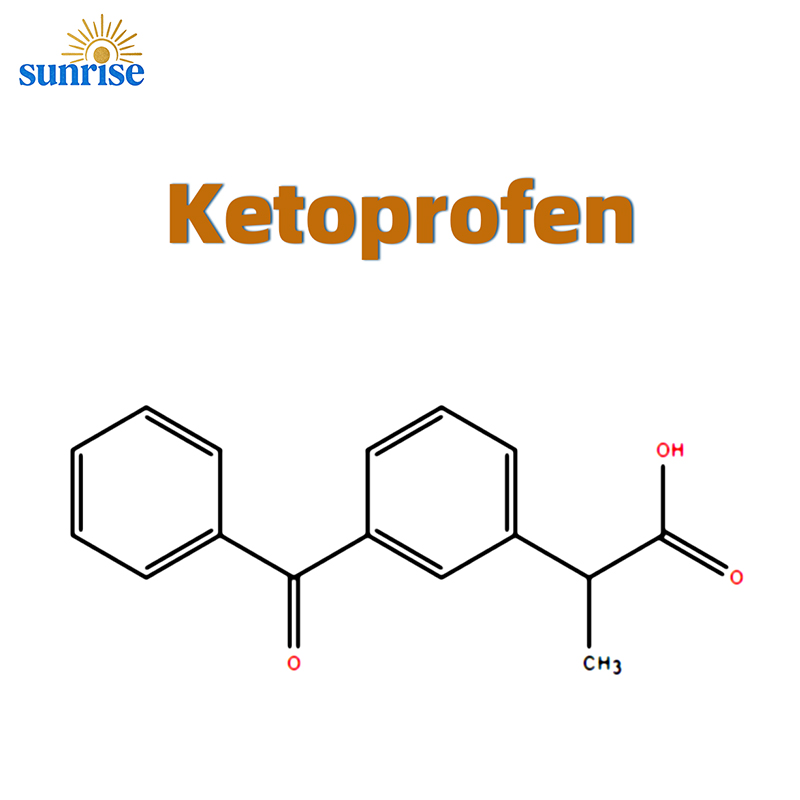
Ketoprofen API CAS 22071-15-4
Ketoprofen,Ketoprofen API,CAS 22071-15-4,Ketoprofen Powder,Ketoprofen USP Standard,Ketoprofen EP Standard,99% Purity Ketoprofen,Micronized Ketoprofen Powder,Ketoprofen Manufacturers,Ketoprofen Bulk Price,Ketoprofen Supplier China,Buy Ketoprofen API,Ketopr
Shaanxi Sunrise offers high-purity (≥99.5%) Ketoprofen API (USP/EP) for global pharmaceutical supply. GMP certified facility. Get a bulk quote and COA/DMF documentation today
Inventory Status:
In Stock & Global Shipping Ready
Click thumbnails to switch | Click image to view HD original
Ketoprofen
CAS Number: 22071-15-4
⚠ Regulatory & Safety Compliance
This Ketoprofen API is manufactured in a GMP-certified facility and is intended for pharmaceutical synthesis, formulation research, and commercial manufacturing. MSDS and COA are mandatory for all shipments.
Ketoprofen: Advanced Pharmacological Insight
Ketoprofen (CAS 22071-15-4) is a propionic acid derivative and a highly potent non-steroidal anti-inflammatory drug (NSAID). Unlike many other analgesics, Ketoprofen exhibits a dual mechanism of action: it effectively inhibits the activity of Cyclooxygenase (COX-1 and COX-2) while also stabilizing lysosomal membranes and antagonizing the effects of bradykinin. This makes it exceptionally effective for managing acute pain and chronic inflammatory conditions like rheumatoid arthritis.
At Shaanxi Sunrise Pharmaceutical, we leverage specialized crystallization technology to produce Ketoprofen API with a superior impurity profile. Our production capacity of 35,000 MT per year ensures price stability and continuous supply for global manufacturers. Our material is optimized for high-speed tablet compression and topical gel formulations, where uniform particle distribution and solubility are critical for bioavailability.
Technical Specifications (Batch COA Summary)
| Quality Indicators | Standard Specification | Analysis Result |
|---|---|---|
| Assay (HPLC) | 99.0% ~ 101.0% | 99.62% |
| Melting Point | 92℃ ~ 96℃ | 94.2℃ |
| Related Substances | Total Impurities ≤ 0.2% | 0.08% |
| Loss on Drying | ≤ 0.5% | 0.12% |
| Heavy Metals | ≤ 20 ppm | ≤ 10 ppm |
Ketoprofen Supply Chain & Support
Global Logistics
Strategic warehouse in Shaanxi allows for rapid dispatch. We support CIF, FOB, and DDP terms with hazardous material-compliant shipping protocols.
Regulatory Dossier
Accelerate your product launch with our comprehensive regulatory package including DMF, MSDS, and Stability Testing data.
Custom Solutions
Special micronized Ketoprofen grades are available upon request to enhance dissolution rates in oral suspension or pediatric formulations.
Standard Packaging
• 25kg Net weight per Fiber Drum
• Double Polyethylene inner liners
• Vacuum packaging available for sensitive R&D batches
Documentation Center
Full COA, Batch Method of Analysis, and Safety Data Sheets are available for instant request.
Request Technical Documents →Shaanxi Sunrise Pharmaceutical Co., Ltd.
Email: betty@pharma-sunrise.com | WhatsApp: +86 18092760627
Leading Supplier of High-Purity Analgesic APIs & Phytochemicals
[xss_clean]function changeView(element, imageUrl) { const viewer = document.getElementById('mainViewer'); if (!viewer) return; viewer.style.opacity = '0'; setTimeout(() => { viewer.src = imageUrl; viewer.style.opacity = '1'; }, 150); const thumbs = element.parentElement.children; for (let thumb of thumbs) { thumb.style.borderColor = '#f1f5f9'; } element.style.borderColor = '#004a99'; }[xss_clean]