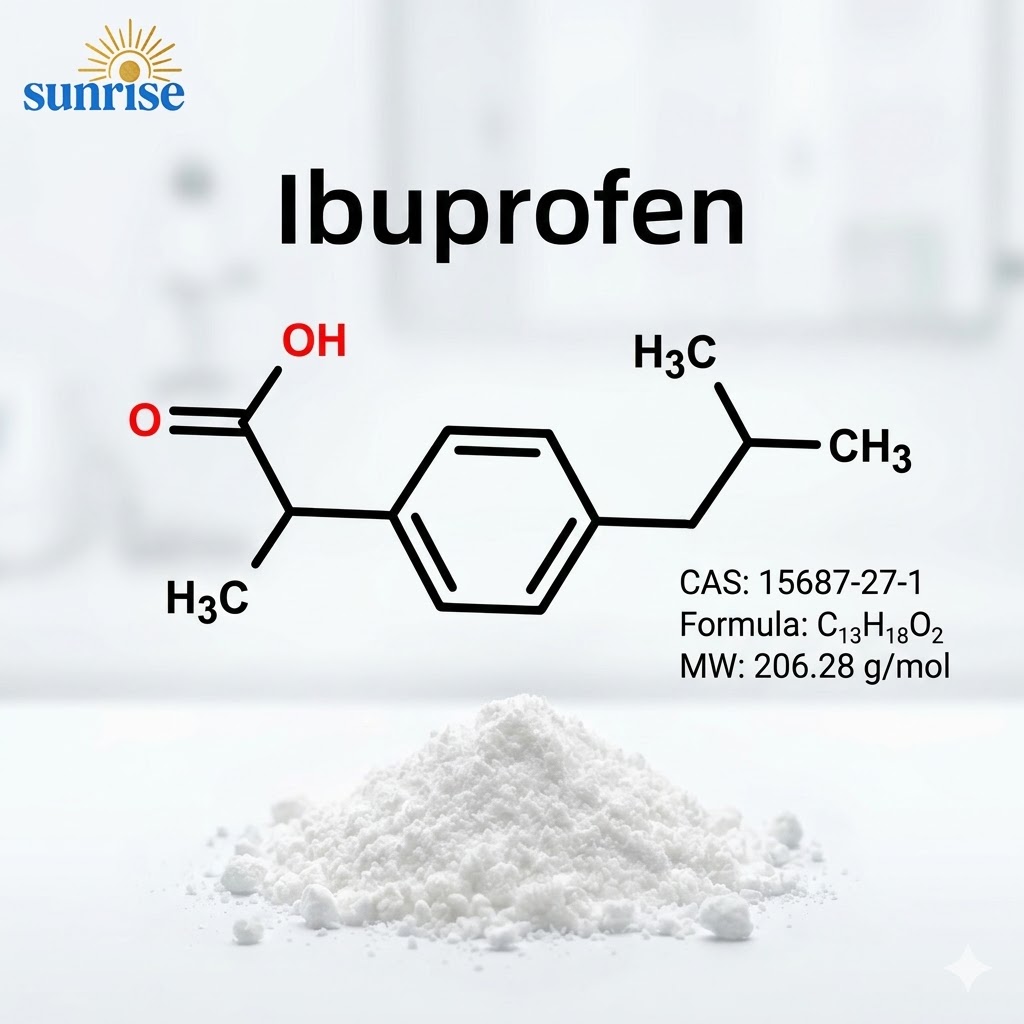
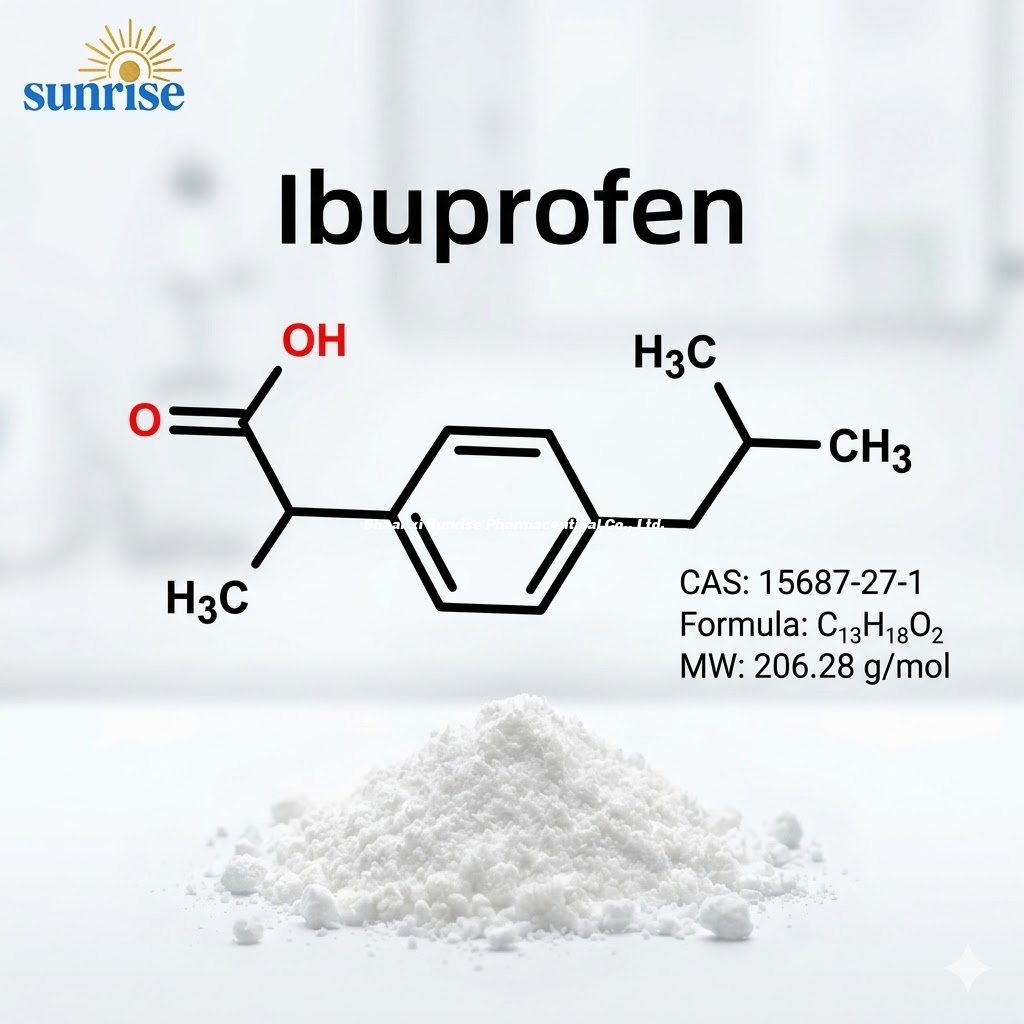
Ibuprofen API CAS 15687-27-1 | High Purity ≥99.5% USP/EP Standard
Ibuprofen API,CAS 15687-27-1,Ibuprofen Powder Bulk,Pharmaceutical Grade Ibuprofen,Propionic acid derivative,NSAID Raw Material,Ibuprofen USP/EP Standard,White Crystalline Powder Ibuprofen,Ibuprofen Manufacturer Shaanxi,Wholesale Ibuprofen Powder,Ibuprofen
Buy high-purity Ibuprofen API (CAS 15687-27-1) ≥99.5% from Shaanxi Sunrise Pharmaceutical. GMP-certified, USP/EP compliant white crystalline powder for fever and inflammation R&D. Bulk supply and worldwide shipping. Request a COA today
Global Export Status:
Ready for Immediate Mass Shipment
Click thumbnails to switch | Click image to view HD original
Ibuprofen (Pure Powder)
CAS No.: 15687-27-1
⚠ Research & Formulation Use
This API is intended for pharmaceutical manufacturing, pharmacological research, and analytical validation. Not for direct human use as a finished medicine.
Scientific & Commercial Overview
Ibuprofen (CAS 15687-27-1) is a non-steroidal anti-inflammatory drug (NSAID) that functions by inhibiting the cyclooxygenase (COX) enzymes, thereby reducing the synthesis of prostaglandins. As a cornerstone of the global analgesic market, high-quality Ibuprofen API is essential for formulating oral tablets, capsules, and topical gels.
At Shaanxi Sunrise Pharmaceutical, we operate at a massive industrial scale with an annual capacity of 50,000 MT. Our Ibuprofen is manufactured using advanced catalytic technology, ensuring a purity profile exceeding 99.5% with ultra-low residual solvents. We provide multiple particle size options, from standard crystalline to micronized powder, to meet diverse formulation requirements in modern pharmaceutical manufacturing.
Technical Specifications (USP/EP Standards)
| Test Parameters | Specification Limits | Typical Batch Result |
|---|---|---|
| Assay (By HPLC) | 97.0% ~ 103.0% | 99.85% |
| Melting Point | 75.0℃ ~ 78.0℃ | 76.2℃ |
| Residue on Ignition | ≤ 0.1% | 0.04% |
| Heavy Metals | ≤ 10 ppm | Conforms |
| Individual Impurity | ≤ 0.1% | 0.02% |
Ibuprofen FAQ & Supply Chain Support
Q1: What documentation can Shaanxi Sunrise provide for pharmaceutical audits?
We provide full regulatory support including DMF (Drug Master File), batch-specific COA, MSDS, Stability Data reports, and GMP compliance certificates to facilitate your local health authority registration.
Q2: Do you offer customized particle size (Mesh) for Ibuprofen?
Yes. We understand that solubility and compressibility are key to your formulation. We can supply Ibuprofen in 40 mesh, 80 mesh, and micronized grades (100 mesh+) to suit your direct compression or granulation processes.
Q3: How do you manage the logistics for 50,000 MT annual capacity?
Our massive production scale is backed by a robust global logistics network. We offer 20ft/40ft container shipping, ISO tank options, and door-to-door DDP services to ensure a stable and cost-effective supply chain for global distributors.
Packaging & Storage
• Bulk Package: 25kg/Fiber Drum with double PE liners
• Custom Packing: Available for 1kg - 10kg R&D samples
• Storage: Store in a cool, dry area. Avoid direct sunlight.
Ready to Scale Up?
Leverage our massive production capacity for your long-term supply security. Contact our B2B team for bulk pricing.
Request Wholesale Price List →Shaanxi Sunrise Pharmaceutical Co., Ltd.
Email: betty@pharma-sunrise.com | WhatsApp: +86 18092760627