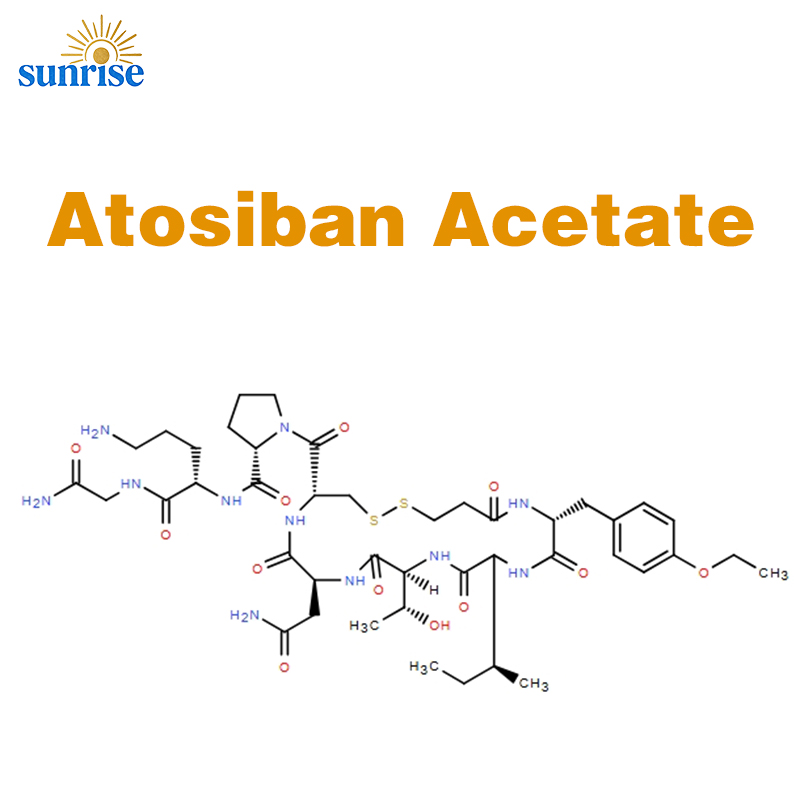
Atosiban Acetate Powder CAS No:90779-69-4
Atosiban Acetate,Atosiban Acetate ≥ 98%,CAS No:90779-69-4
Product Name: Atosiban Acetate API Powder
Product content: ≥ 98%
CAS No:90779-69-4
Atosiban Acetate (API)
High-Purity Peptide for Obstetric Research and Preterm Labor Management
This product is intended for Industrial Use or Scientific Research ONLY. Shaanxi Sunrise Pharmaceutical provides high-specification Atosiban Acetate raw materials (≥99% purity available) under strict pharmaceutical quality control systems.
Product Description
Shaanxi Sunrise Pharmaceutical specializes in high-quality peptide synthesis. Our Atosiban Acetate is a synthetic cyclic nonapeptide that accurately meets the stringent requirements of pharmaceutical R&D. Characterized by its excellent water solubility and batch-to-batch consistency, this API provides a reliable foundation for formulating uterine contraction inhibitors.
Mechanism of Action (MOA)
Selective Receptor Antagonism
Atosiban Acetate acts as a competitive antagonist against key receptors in the uterine smooth muscle:
OXTR & V1AR Binding: Competitively binds to Oxytocin Receptors (OXTR) and Vasopressin V1A Receptors, blocking contraction signaling.
Calcium Channel Inhibition: Reduces intracellular calcium release and extracellular calcium influx, leading to smooth muscle relaxation.
Rapid Onset: Clinically proven to reduce contraction frequency and intensity within 10 minutes of administration (research reference).
Applications in Research
Preterm Birth Delay: Study of therapeutic windows for fetal lung maturation.
Assisted Reproductive Technology (ART): Research into reducing uterine activity during embryo transfer.
Comparative Pharmacodynamics: Benchmarking against traditional inhibitors like β-agonists for superior cardiovascular safety.
Toxicological & Safety Profile
● Organ Safety: No organ pathological damage observed in 3-month high-dose animal studies (10x human dose).
● Reproductive Integrity: No evidence of fetal malformation, genetic mutation, or carcinogenicity in standard Ames/Chromosomal tests.
Frequently Asked Questions (FAQ)
What is the difference between CAS 90779-69-4 and 914453-95-5?
CAS 90779-69-4 refers to the Atosiban base, while 914453-95-5 refers to the Acetate form. Our product is the high-purity Acetate form, which is the standard used in pharmaceutical injectables.
How do you handle the high water solubility of Atosiban during storage?
While its solubility (≥50 mg/mL) is an advantage for formulation, it makes the powder hygroscopic. We use triple-layer vacuum sealing and recommend storage in a dry, dark environment at -20°C to prevent moisture absorption.
Is this API suitable for ART research?
Yes. Atosiban is a hot topic in Assisted Reproductive Technology for improving implantation rates by maintaining uterine stillness. Our ≥99% purity grade is ideal for these sensitive research applications.
What documentation is provided for batch consistency?
We provide a full COA, HPLC chromatogram, and Mass Spectrometry (MS) report for every batch to ensure you receive a consistent cyclic peptide structure.