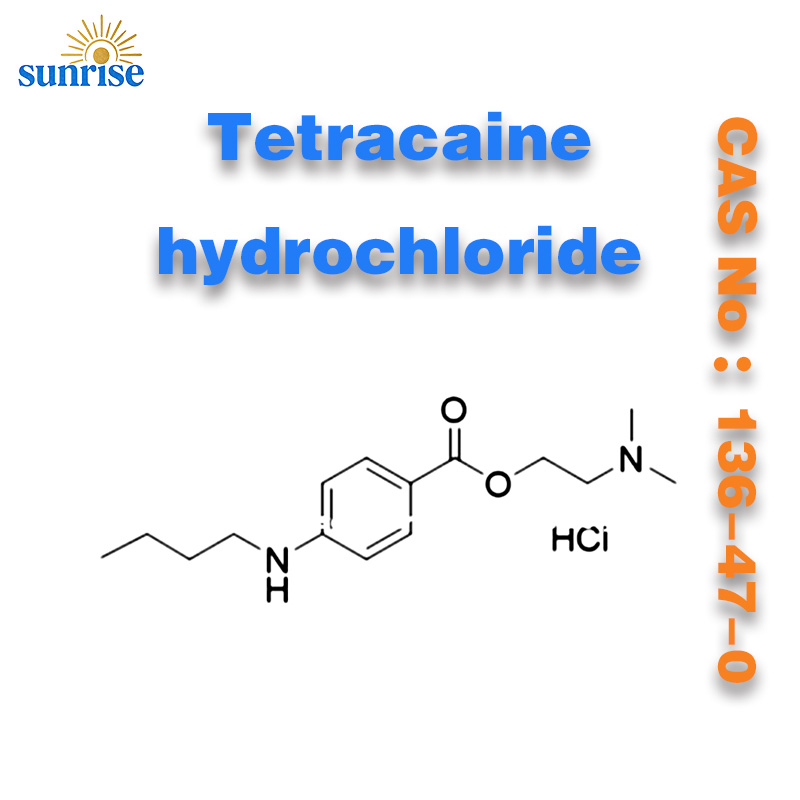
Tetracaine hydrochloride CAS No: 136-47-0
Tetracaine hydrochloride ,136-47-0, Tetracaine hydrochloride CAS No: 136-47-0
Product Name: Tetracaine hydrochloride
Product Purity: ≥ 99%
CAS No :136-47-0
Tetracaine Hydrochloride Powder

Store in a dry, cool (2-8°C recommended), and light-proof warehouse. Keep container tightly sealed to prevent moisture absorption and maintain 24-month stability.
Product Description
Tetracaine Hydrochloride by Shaanxi Sunrise Pharmaceutical is a long-acting ester-type local anesthetic API. It provides rapid onset and prolonged duration by effectively blocking voltage-gated sodium channels. Our product is manufactured in a Class D cleanroom environment, ensuring full compliance with international pharmacopoeias for use in high-end ophthalmic and surgical formulations.
| CAS Number | 136-47-0 |
| Molecular Formula | C₁₅H₂₅ClN₂O₂ |
| Molecular Weight | 300.82 g/mol |
| Assay (Dried Basis) | 99.0% - 101.0% |
| Appearance | White crystalline powder |
| Standard Pack | 1 kg/bag; 25 kg/drum |
| Shelf Life | 24 Months |
Unique Advantages
Superior Quality Standards
Purity ≥99% with total impurities ≤0.5%. Every batch is strictly tested via HPLC/GC to ensure consistency and patient safety.
Optimized Synthesis
Utilizing high-efficiency catalysts, our production process ensures high yield (>97%) and extremely low residual solvents (ICH Q3C compliant).
Global Compliance
Produced in GMP-certified facilities with full traceability and detailed COA, supporting FDA/EMA regulatory submissions.
Mechanism of Action
Tetracaine Hydrochloride exerts its therapeutic effect through reversible neural blockade:
Sodium Channel Blockade: Inhibits sodium ion influx on nerve membranes, preventing action potential generation.
Prolonged Duration: Higher lipophilicity allows for deeper tissue penetration and 2-3 times longer effect than procaine.
Wide Application: Primary active ingredient for ophthalmic drops, ENT sprays, and spinal anesthesia injections.
Technical Support & FAQ
Does it comply with USP/EP standards?
What are the available packaging options?
Can you provide customized particle sizes?
⚠ Safety Warning & Declaration
This product is a pharmaceutical raw material intended for industrial manufacturing and laboratory research only. It is not for direct human use. Shaanxi Sunrise Pharmaceutical assumes no liability for unauthorized handling.