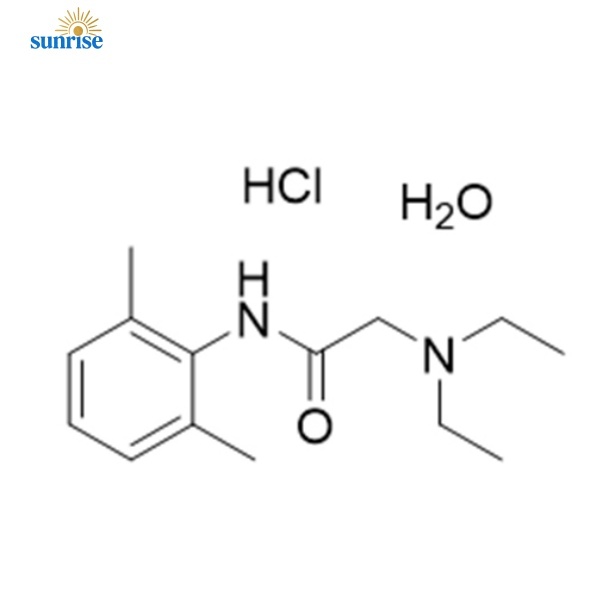
Lidocaine hydrochloride CAS No:73-78-9
Lidocaine hydrochloride, CAS 73-78-9 ,Local anesthesia,Lidocaine hydrochloride CAS No:73-78-9
Product Name: Lidocaine hydrochloride
Product Purity: ≥ 99.0%
CAS No :73-78-9
Lidocaine Hydrochloride Powder

Highly soluble in water and ethanol. Store in a dry and cool place, protected from direct sunlight and high temperatures. Shelf life is 24 months in sealed containers.
Product Introduction
Lidocaine Hydrochloride is a high-purity pharmaceutical-grade amide-type local anesthetic and Class Ib antiarrhythmic API. Synthesized via a refined GMP-compliant process, it provides consistent pharmacological activity for injectable, topical, and mucosal formulations. Our product ensures extremely low impurity profiles, meeting the highest global regulatory expectations.
| CAS Number | 73-78-9 |
| Chemical Name | 2-(diethylamino)-N-(2,6-dimethylphenyl)acetamide HCl |
| Molecular Formula | C₁₄H₂₃ClN₂O (Anhydrous) |
| Molecular Weight | 270.80 |
| Appearance | White crystalline powder |
| Standard Pack | 1 kg/bag; 25 kg/drum |
| Shelf Life | 24 Months |
Product Advantages
Pharmacopoeia Purity
Assay ≥99.5%. Meets USP43, EP11, and BP requirements. Total related substances ≤0.5% and heavy metals ≤10 ppm for maximum clinical safety.
Scale Production
100,000-level clean production workshop with an annual output exceeding 300 tons, ensuring reliable supply for global partners.
Regulatory Support
Full documentation package including batch-specific COA, MSDS, GMP certificate, and DMF assistance for major global markets.
Typical Certificate of Analysis (COA)
| Test Item | Specification | Result | Method |
|---|---|---|---|
| Appearance | White crystalline powder | Conforms | Visual |
| Identification | Matches IR Spectrum | Conforms | USP 43 <197K> |
| Assay (HPLC) | 99.0% – 101.0% | 99.5% | Chromatography |
| pH (1% Solution) | 4.0 – 5.5 | 4.8 | USP 43 <791> |
| Loss on Drying | ≤1.0% (Monohydrate) | 0.45% | 105℃ for 2 hrs |
| Residue on Ignition | ≤0.1% | 0.04% | USP 43 <281> |
⚠ Safety Warning & Usage
Note: The anhydrous form (MW 270.80) is less stable; the monohydrate form is the industry standard for medicinal applications. This product is a pharmaceutical raw material and should only be handled by qualified professionals in GMP-regulated environments.