Dapagliflozin Raw Powder API CAS No: 960404-48-2
Dapagliflozin,960404-48-2,Dapagliflozin Raw Powder API CAS No: 960404-48-2,Dapagliflozin Raw Powder API,CAS No: 960404-48-2
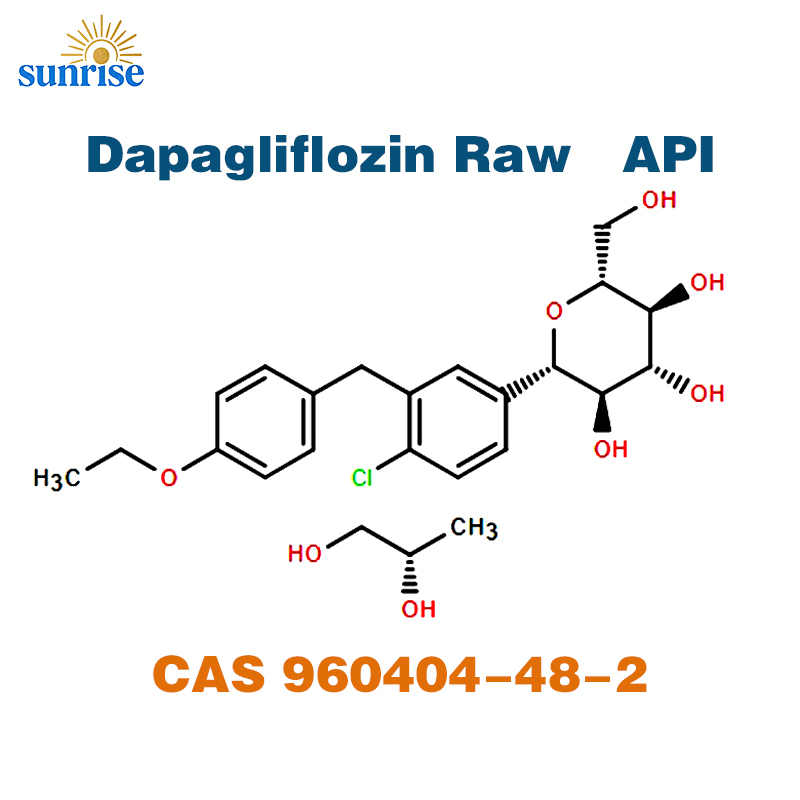
Dapagliflozin Raw Powder API

Store at -20°C for long-term stability. Stock solutions should be kept at -80°C to maintain maximum potency for research applications.
Product Description
Dapagliflozin is a highly selective Sodium-Glucose Co-transporter 2 (SGLT2) inhibitor. This high-purity API is a C-glycosyl compound characterized by its glucose sugar unit linked to an ethoxybenzyl-chlorobenzene moiety. Produced under rigorous quality standards, it is optimized for oral solid dosage formulations (tablets/capsules).
| CAS Number | 960404-48-2 |
| Molecular Formula | C₂₁H₂₅ClO₆ |
| Molecular Weight | 408.87 g/mol |
| EINECS No. | 811-335-4 |
| Appearance | White to off-white crystalline powder |
| Standard Pack | 1KG/Foil Bag; 25KG/Drum |
| Shelf Life | 24 Months |
Core Advantages
Insulin-Independent
Works via the renal glucose excretion pathway, remaining effective regardless of the patient's insulin resistance levels.
Industrial Stability
The crystalline form ensures consistent flow properties and optimal solubility during industrial tableting and encapsulation.
Global Compliance
Minimized impurity profile (Purity ≥99.0%) ensures maximum safety for finished drug product manufacturing.
Mechanism of Action
Dapagliflozin provides multi-organ therapeutic benefits through three core functions:
1. Glucose Regulation: Inhibits SGLT2 in renal tubules, reducing reabsorption and increasing urinary glucose excretion.
2. Cardio-Renal Protection: Reduces cardiac workload and intraglomerular pressure, aiding Heart Failure and CKD management.
3. Osmotic Diuresis: Assists in blood pressure reduction by reducing intravascular volume through glucose-driven diuresis.
Technical Support & FAQ
What is the purity standard for this API?
Which formulations is this API suitable for?
Do you provide technical documentation?
⚠ Safety Warning & Declaration
This product is a chemical raw material intended for professional laboratory research or pharmaceutical manufacturing only. It is not for direct human consumption. Shaanxi Sunrise Pharmaceutical Co., Ltd. assumes no liability for improper handling.